 
Rubidium and caesium are denser than water. 
Lithium, sodium and potassium are less dense than water and therefore can float on water. Similarly, on the application of heat the free moving electrons in the outermost shells gain a lot of energy and transfer this through the metal via collisions with the other free moving electrons. On the application of an electric field the large numbers of free flowing electrons in the outer shells conduct electricity through the metal. The outer electron can drift further from the nucleus and move relatively freely. It is the reduced interatomic forces in these elements that make them relatively soft.Īlkali metals are extremely good conductors of electricity and heatĪlkali metals have one electron in their outermost shell which is held very weakly by the nucleus.  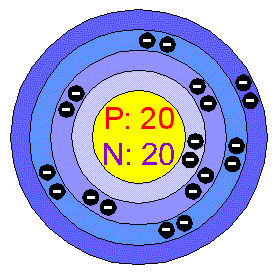
Freshly cut alkali metals are shiny but tarnish rapidly due to reaction with oxygen in the air. Alkali Metals are softĪlkali metals are soft metals that can be cut with a knife and silvery coloured. Sections below cover the trends in atomic radius, first ionization energy, electronegativity, melting and boiling points, and density. The increasing atomic radius means weaker forces between the atoms and so a lower melting and boiling point. Jim Clark Truro School in Cornwall This page discusses the trends in some atomic and physical properties of the Group 1 elements - lithium, sodium, potassium, rubidium and cesium. Caesium also has the highest electropositivity of all known elements and fluorine has the highest electronegativity of all known elements. Caesium fluoride can be used in organic synthesis as a source of the fluoride anion. The decrease in melting and boiling points down the group can be explained by the additional shell being added to the previous element causing the atomic radius to increase. Caesium fluoride or cesium fluoride is an inorganic compound with the formula CsF and it is a hygroscopic white salt. Cesium was discovered spectroscopically in 1860 by Bunsen and Kirchhoff in mineral water from Durkheim. It is these weaker attractive forces due to the large atomic radii between neighbouring atoms of Group 1 elements that result in lower melting and boiling points when compared to other metals. Melting Point: 28.5 ☌: Atomic Weight: 132.9: Boiling Point: 671 ☌: Electron Configuration: Xe6s 1: Oxidation States: +1, 1 (a strongly basic oxide) History. The large atomic size results in weaker forces between neighbouring atoms. This results in Group 1 elements having larger atomic radii than those elements that follow them in their respective periods. This electron can drift further from the nucleus than in most atoms of other elements. ElementĪlkali Metals have lower melting and boiling PointsĪll Group 1 elements have one electron in their outermost shell which is held very weakly by the nucleus. Minimize contact.The table below summarises the physical properties of the Group 1 elements. It is a soft, silvery-golden alkali metal with a melting point of 28.5 ☌ (83.3 ☏), which makes it one of only five elemental metals that are liquid at or near room temperature. ORL-RAT LD50 2390 mg kg-1, IVN-RAT LD50 1210 mg kg-1, SCU-MUS LD50 2200 mg kg-1 OU Chemical Safety Data (No longer updated) More details Toxicity: Inorganic Compound Nitrite Nitrate Industrial/Workplace Toxin Synthetic Compound Toxin, Toxin-Target Database T3D3553.OU Chemical Safety Data (No longer updated) More details Incompatible with strong reducing agents, alcohols, organicmaterials, aluminium. Contact with combustible material may cause fire.Decomposes at high temperatures. Appearance: colourless crystals OU Chemical Safety Data (No longer updated) More details.Several binary (containing only Cs and O) oxides of caesium are known. Experimental Density: 3.68 g/mL Alfa Aesar 12884, 14440, 38617ģ.685 g/mL Parchem – fine & specialty chemicals 31122 Caesium oxide ( IUPAC name), or cesium oxide, describes inorganic compounds composed of caesium and oxygen. 5 The structure of copper is described as a lattice of positive ions in a ‘sea of electrons’.Slightly soluble in alcohol Alfa Aesar 12884 Experimental Solubility: Soluble in 5 parts cold and 0.5 parts boiling water.Experimental Refraction Index: 1.55 Alfa Aesar 38617, 14440, 12884ġ.55 Parchem – fine & specialty chemicals 31122.Experimental Boiling Point: 233 ☌ Parchem – fine & specialty chemicals 31122.Experimental Melting Point: 414 ☌ Alfa AesarĤ14 ☌ OU Chemical Safety Data (No longer updated) More detailsĤ14 ☌ (Literature) Alfa Aesar 12884, 14440, 38617Ĥ14 ☌ Parchem – fine & specialty chemicals 31122.Experimental Physico-chemical Properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
